This past summer, I raised honey bees on different diets on the Tufts Vet School campus in Grafton, MA. I am interested in how those different diets may, or may not, have affected the community composition of the honey bee gut microbiome. The honey bee gut is full of bacteria that may be beneficial to honey bee health. To investigate how these bacteria might be influenced by diet, I followed my Tufts Vet fieldwork with some hard-core lab work.
Throughout the summer, my undergraduate interns and I sampled honey bees from our hives on a weekly basis. The sampled bees were chilled, and put in ethanol until it was time for lab work. While I felt torn euthanizing my bees, we only sampled 10 bees per hive per week. In the grand scheme of the hive (which can have tens of thousands of individuals), we weren’t making a dent. And, it was all in the name of science, and better understanding pollinator nutritional ecology!
Once the fieldwork was over (the part I’m used to), the lab work began (the part I’m not used to). Thankfully, I had help from the Wolfe Lab. In September, as summer turned to fall, I dissected the preserved bees. Since I am interested in the microbiome of the hindgut (where most of the bacteria live), I isolated that specific part of the bee. Believe it or not, dissecting the honey bee gut is relatively easy (with some practice). If you’re interested, I followed the procedure from TheBeeVlog’s YouTube video.
Once my honey bee hindguts were isolated, it was time to extract the bacterial DNA. Once the bacterial DNA was extracted, I could use a method, called metabarcoding, to identify which bacteria were living in my honey bee hindguts based on the bacterial DNA sequences! To extract bacterial DNA, I put the dissected guts into “PowerBead Tubes” (i.e. tubes with some liquid and small beads), and shook up the tubes for a while.
It was sort of mesmerizing.
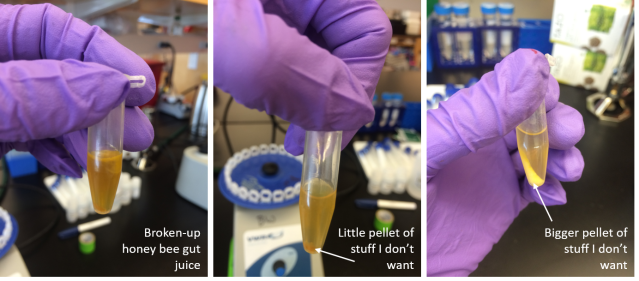
Then, I mixed my broken-up bee guts with various chemical compounds. As I added chemicals and spun down the tubes of broken-up-bee-gut juice, most of the stuff that wasn’t bacterial DNA made a pellet at the bottom of the tube. Then, I took the juice at the top of the tube, where the bacterial DNA is, and added more chemicals until I got nice, clear DNA juice!
Once that was finished, I was ready to clean up my DNA for sequencing! I was on schedule to be done with my lab work by November. But, there was a problem. There were signs of contamination in the Wolfe Lab. Everything came to a halt while the Wolfe Lab ran test after test to figure out what the source of contamination could be. Thankfully, the source of the error was with a certain DNA primer, which I hadn’t used. Phew.

In December, I started up my lab work again. I pipetted more in those four days than in my entire career as a scientist. Hard-core lab work. After boxes and boxes of pipet tips and running gel after gel (to make sure I actually had bacterial DNA), I had done it! I had cleaned up all the bacterial DNA from all my bee guts. I had put hours of work into producing one tiny tube of bacterial DNA (can you see it?).
For someone who is used to field work and macroecology, the whole experience was a bit mind-boggling, but fruitful. I am eternally grateful to the Wolfe Lab members for all their help in this project, and I am glad to have learned lab skills along the way.
Now, I wait for the data. Science can be a lot of waiting, but I can’t wait to see what I found!
Me and my tiny tube of DNA. Photo courtesy of Ben Wolfe.